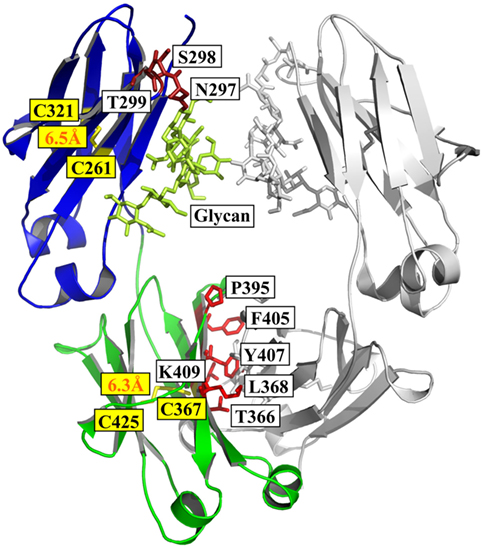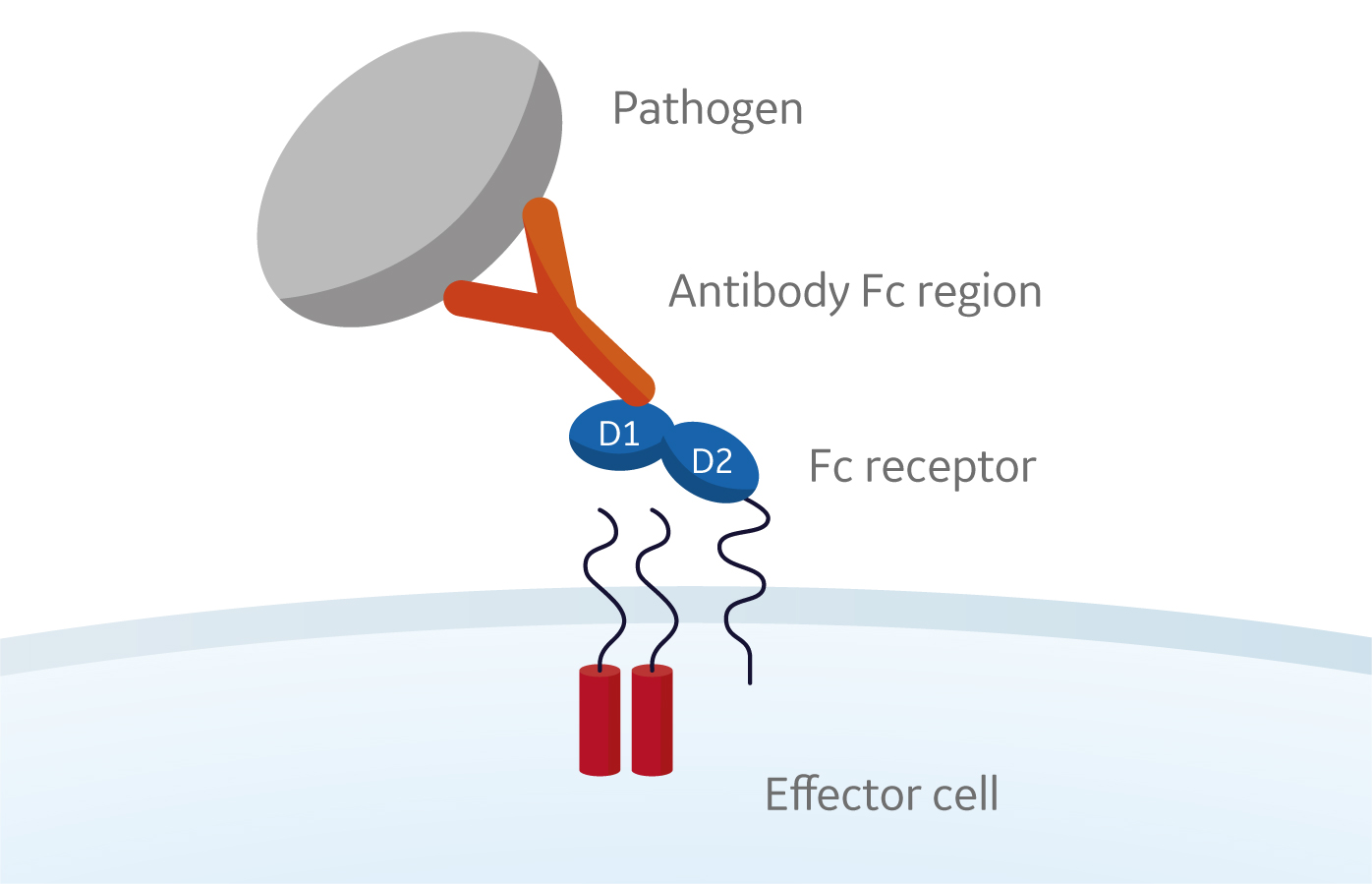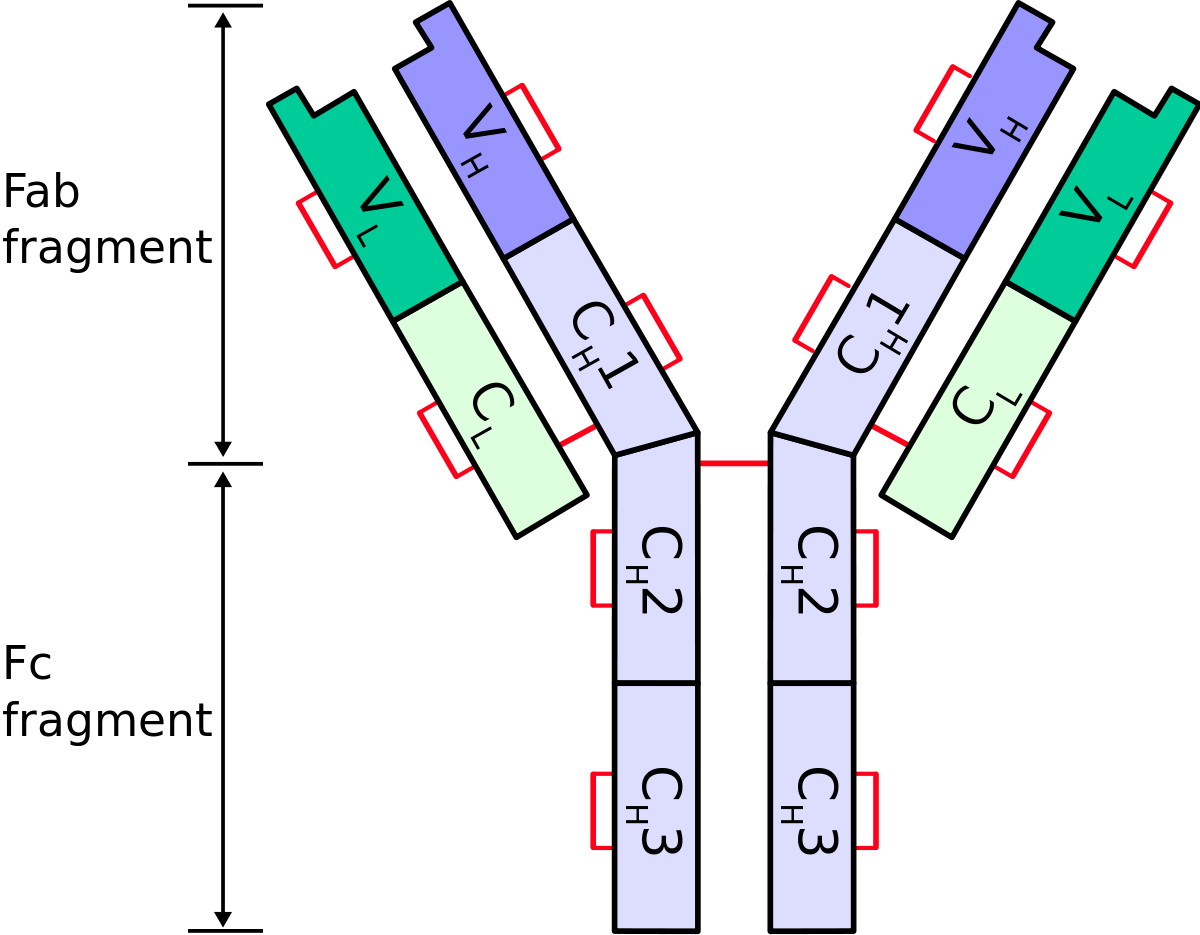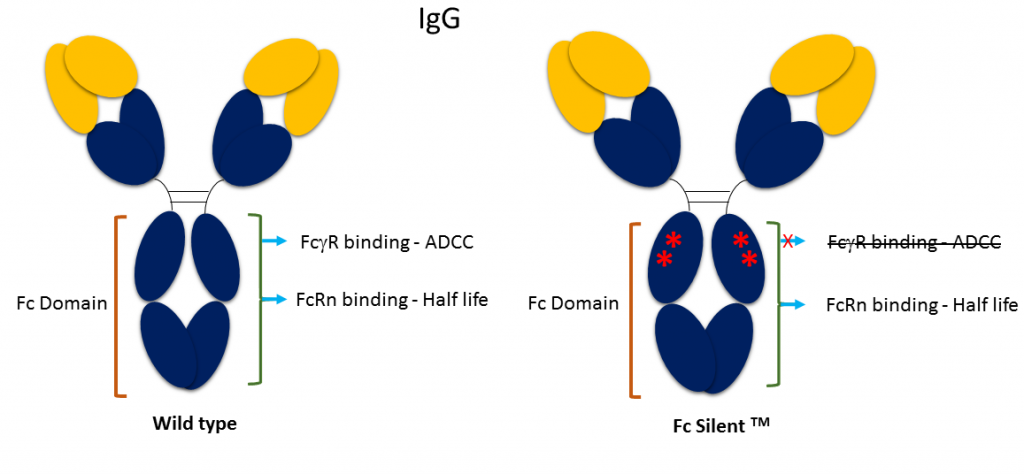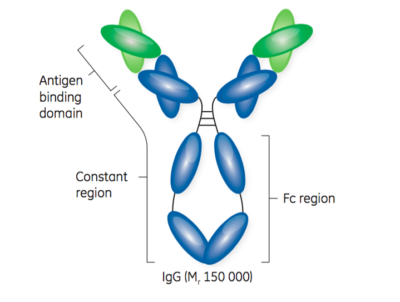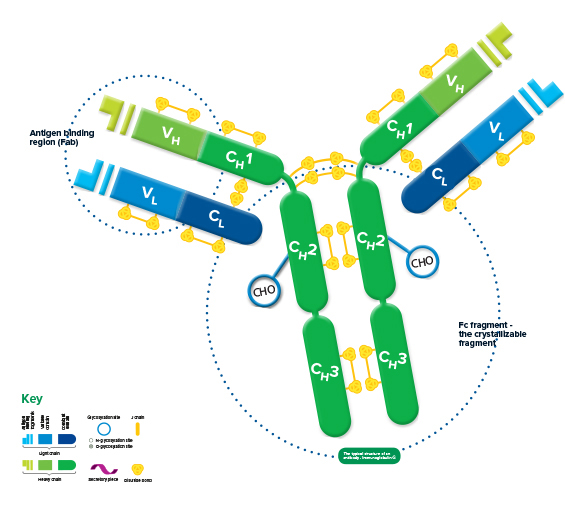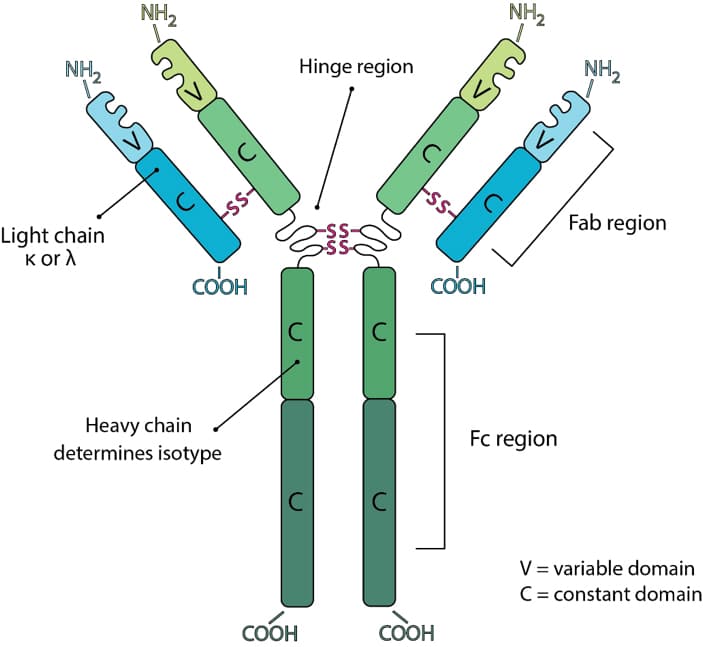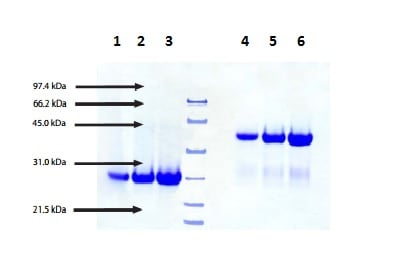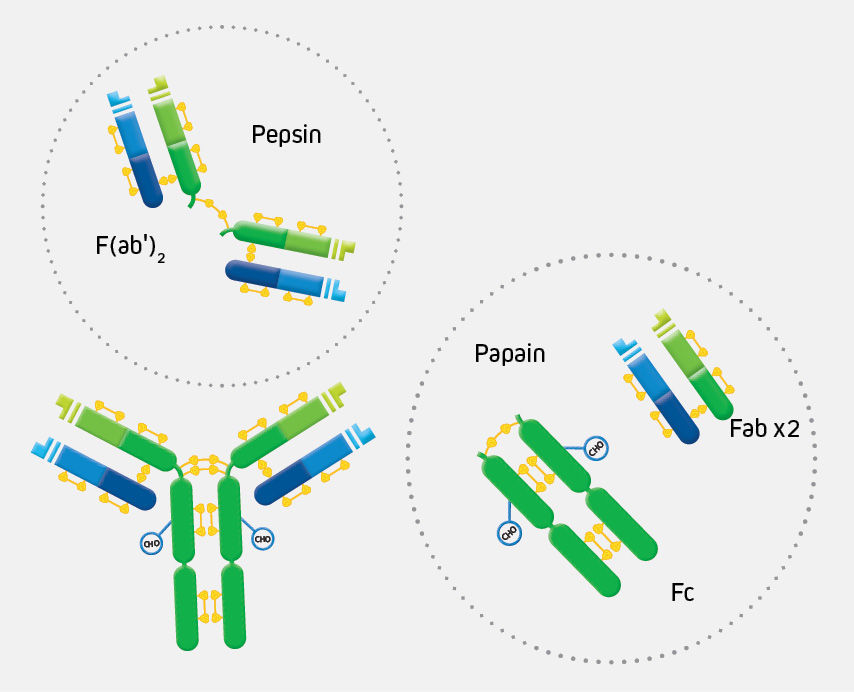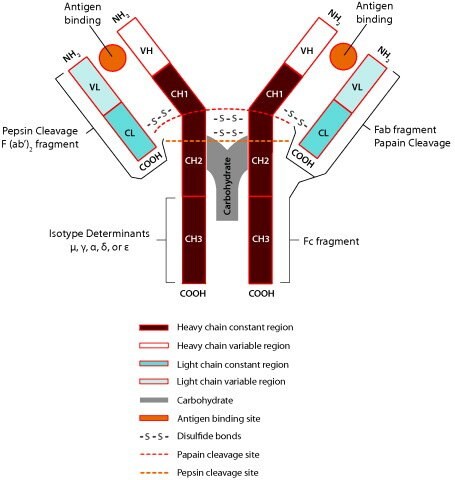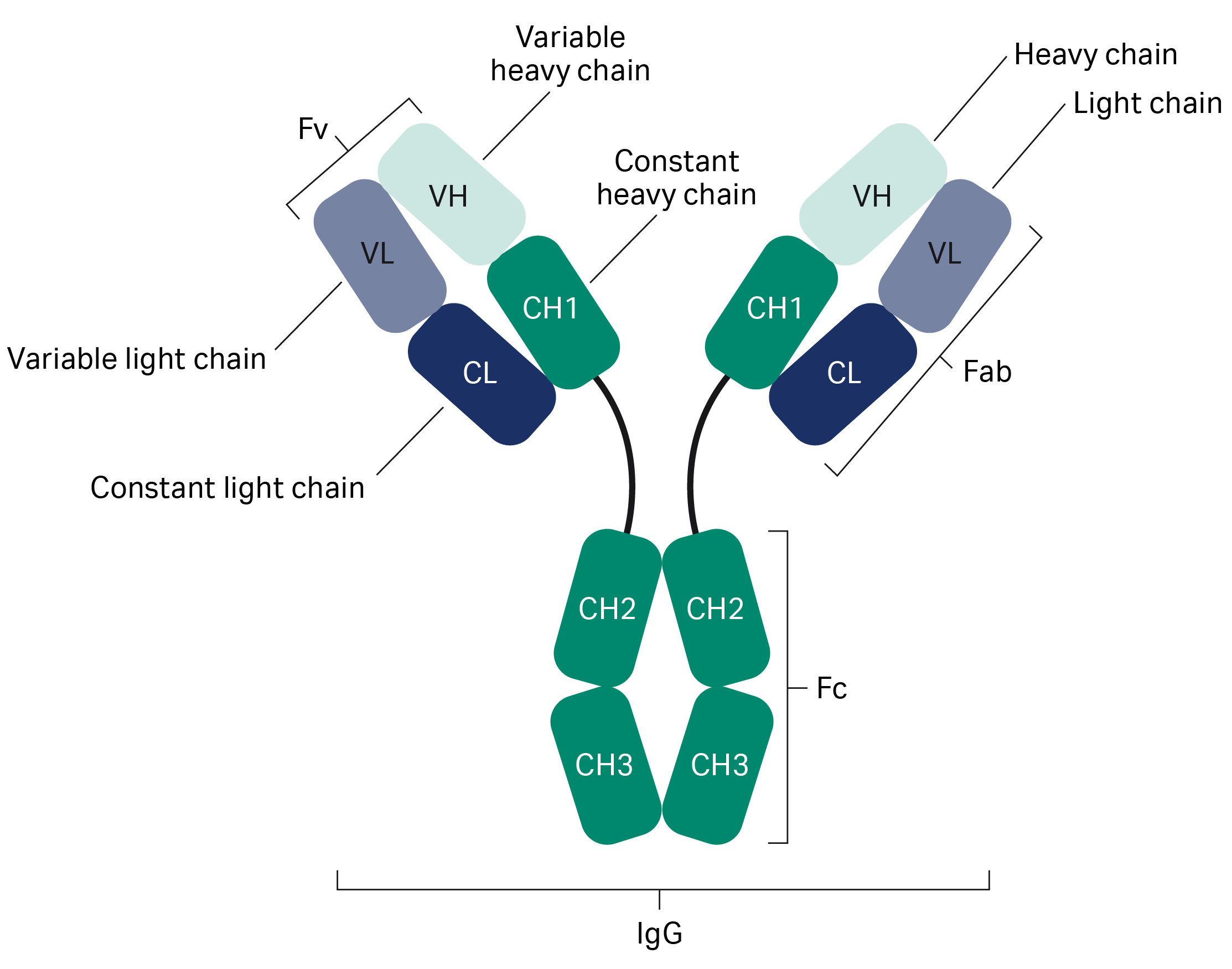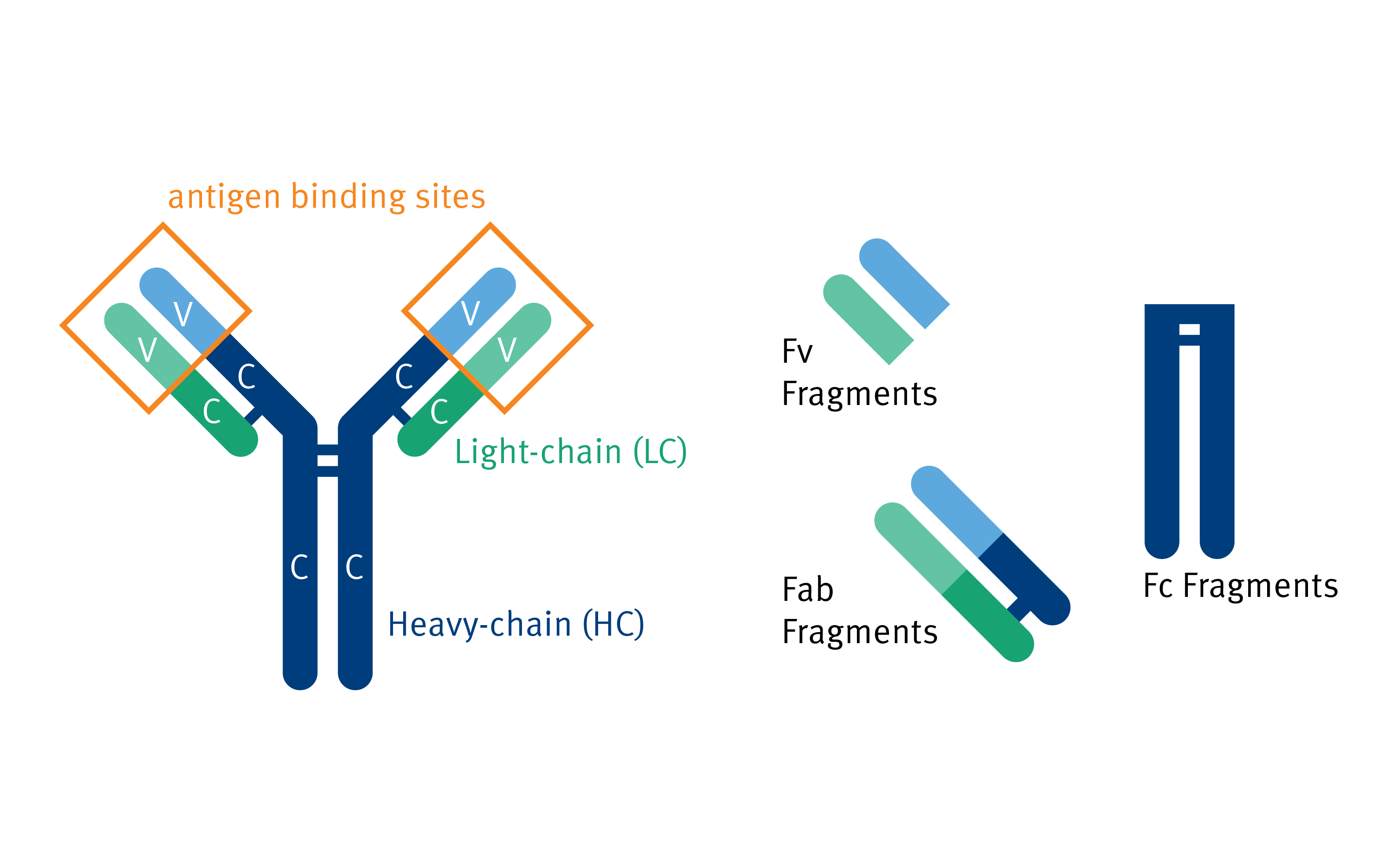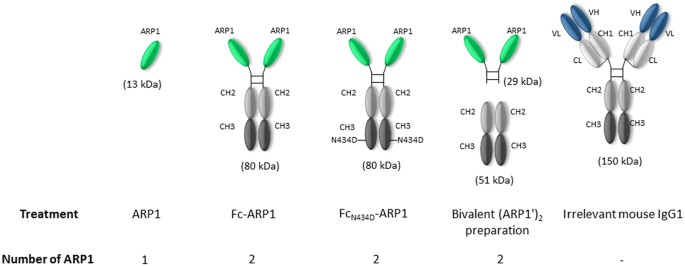
Fusion of the mouse IgG1 Fc domain to the VHH fragment (ARP1) enhances protection in a mouse model of rotavirus | Scientific Reports
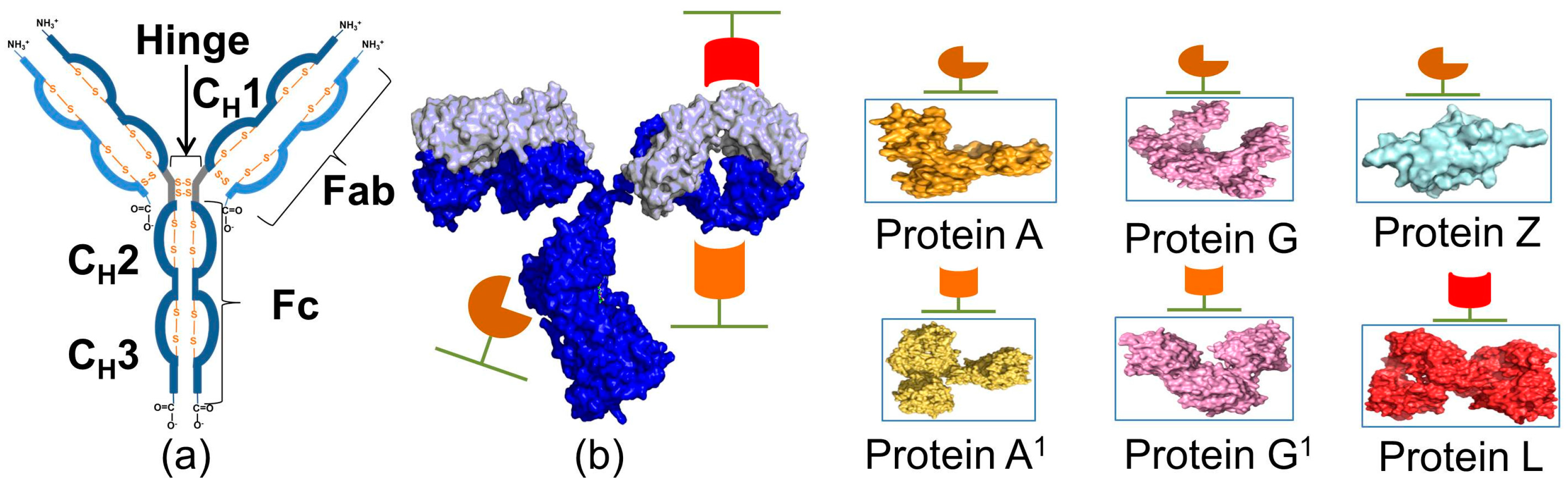
Materials | Free Full-Text | Fc-Binding Ligands of Immunoglobulin G: An Overview of High Affinity Proteins and Peptides
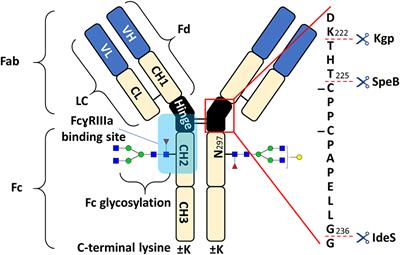
Frontiers | Proteoform-Resolved FcɤRIIIa Binding Assay for Fab Glycosylated Monoclonal Antibodies Achieved by Affinity Chromatography Mass Spectrometry of Fc Moieties

Binding characteristics of staphylococcal protein A and streptococcal protein G for fragment crystallizable portion of human immunoglobulin G - ScienceDirect
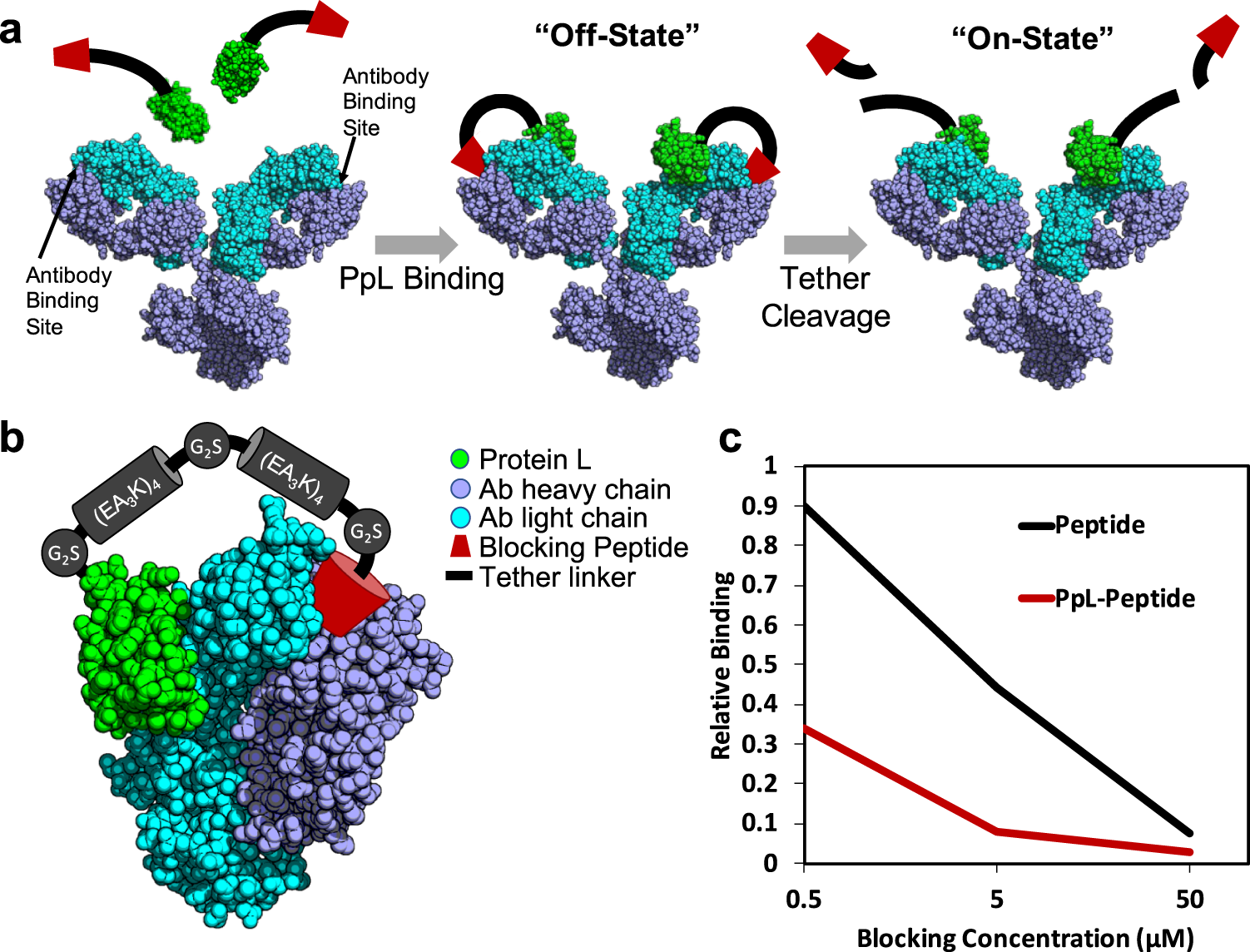
Turning antibodies off and on again using a covalently tethered blocking peptide | Communications Biology
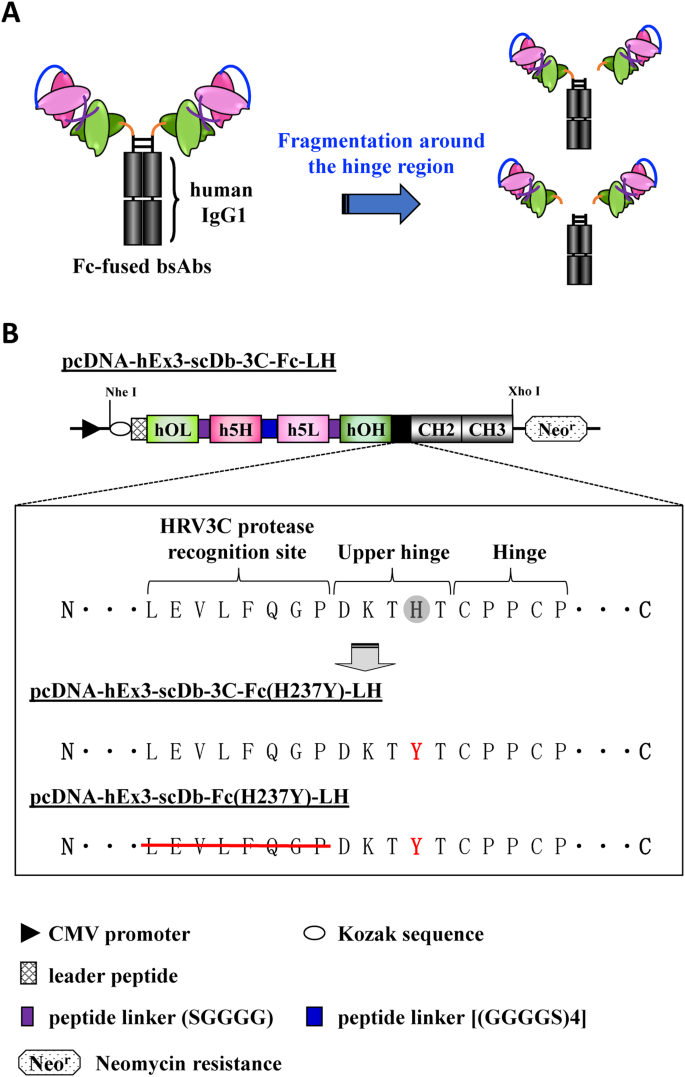
Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports
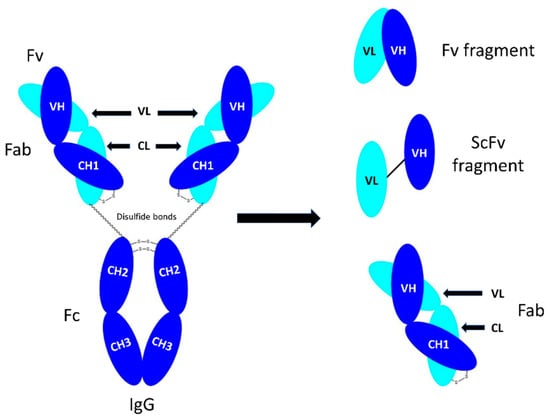
Bioengineering | Free Full-Text | Soluble Papain to Digest Monoclonal Antibodies; Time and Cost-Effective Method to Obtain Fab Fragment