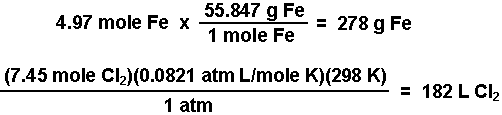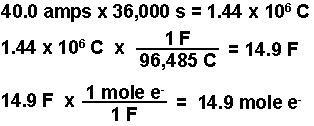Question Video: The Ionic Equation for the Electrolysis of the Molten Salt of Barium at the Negative Electrode | Nagwa

SOLVED: For problems involving calculations you must show your work for credit. Unless otherwise stated,you may assumeT=25.0C 5 Electrolysis is used to cover rings made of inexpensive metals with a coating of
Welcome to Chem Zipper.com......: During electro refining of Cu how much time is needed to produce 250g Cu on the cathode if the current is kept at 11 A?
20. Calculate the quantity of electricity in coulomb which liberates enough hydrogen at the cathode during electrolysis of acidified water so that it can fill a ballon of capacity 10 litres at

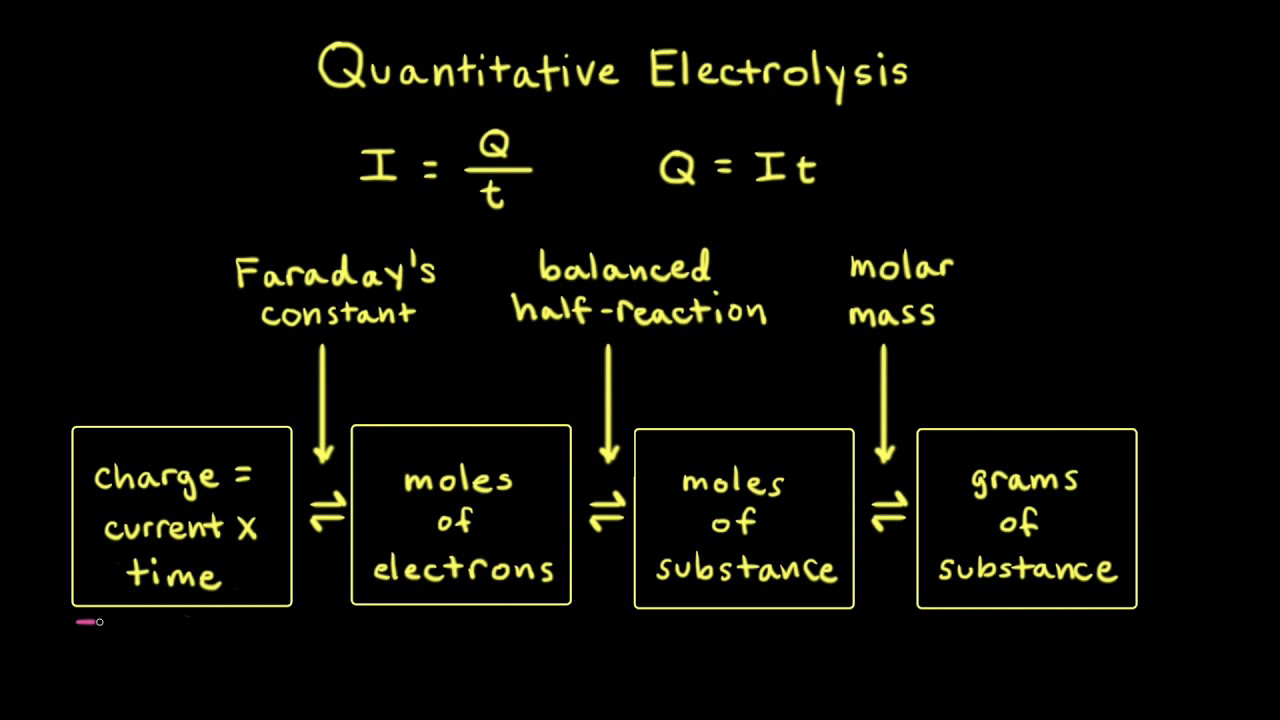
Unit 3 PPA 2 QUANTITATIVE ELECTROLYSIS. QUANTITATIVE ELECTROLYSIS (Unit 3 PPA 2) The aim of this experiment is to determine the quantity of electricity. - ppt download